To make things easier, and assuming you require a rough estimate, you could use a straight density figure of 1g/ml. The source I used did not give temperature so even this is an estimate.īy the way, in the United States we do not use imperial gallons or imperial anything, at lest not since the revolution, we use a scale called avoirdupois I guess the originator of the thread was British? In avoirdupois the measurement is so close as to be meaningless in the home, not so in the Imperial system.īy the way, a pint of beer in the UK measures around 20 Fluid Ounces in the US. Water density at room temperature: 0.99802 g/ml. 
Pure water has a density of 1g/cm 3 or 1000kg/m 3. Third Step (Solution): Now, you substitute your formula for the density of water with the data available to you and solve the problem. The density is defined as the mass per unit volume ratio. Second Step (Formula): In this stage, you state the equation that will help you to calculate the density of water, which is Density () Mass (m) / Volume (v) or m/v. Is the density of water 1 or 997 Water has a density of 997 kg/m 3 at 25 degrees Celsius. Grams or kilograms per litre are also available. The density of a substance is the same regardless of the size of the sample. The output if calculating density is in grams per cm 3 or kg per m 3, as well as in oz/cu in, lb/cu ft and lb per cubic yard. Since Ounces weight (avoirdupois) and Fluid Ounces are actually totally unrelated scales (the term ounce has many meanings) there happens to be a nice accident so that a fluid ounce of water actually weighs 1.0425 ounces avoirdupois. The density of water is 1 gram per cubic centimeter. 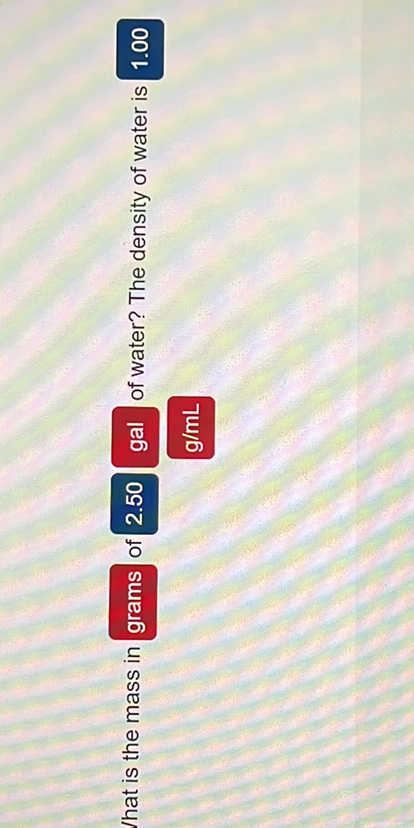
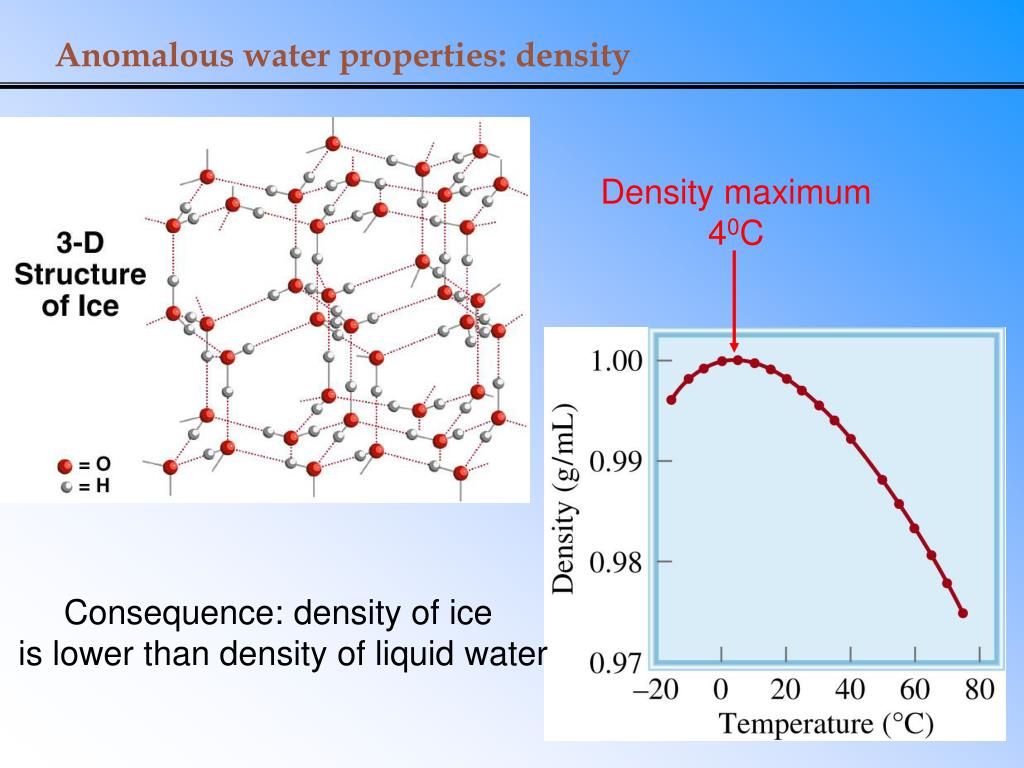
The density of water (H 2 O) changes depending on temperature and pressure - as with other substances. In American units the density of water = 8.32 lbs/gal and hence it is not coincidental that the density cup has a volume of 83.2 cm3 or 0.0832 L.How technical and accurate do we need to be in baking? Here are the exact densities and, yes, temperature does matter, in analytical chemistry, not in baking.ġ milliliter (ml) = 1 cubic centimeter (cc) of pure water at 4 degrees centigrade - water's densist state (it expands on the phase change of freezing into a solid), so, 1 cubic centimeter contains 1 gram of water at 4 deg. Data chart with the density of water at different temperatures. One liter = 1,000 g, and the density of water is 1,000 g/L. Both the blood and urine have dissolved substances in water that increase the density from that of pure water. The density of urine varies in the range of 1.005 to 1.030. One gram is the weight of 1 cm3 of water. For example, the specific gravity of whole blood for humans is 1.060. The definitions of grams and liters are based on the density of water. 
Now let us convert grams to lbs, and liters to gallons: Let the weight of the coating in the density cup = X g Density (g/cm 3) State of Matter hydrogen : 0.00009: gas: helium (at STP) 0.000178: gas: carbon monoxide (at STP) 0.00125: gas: nitrogen (at STP) 0. Calculate the density of (a) a piece of peach whose mass is 37.42 g and which, when submerged in water, increases the water level in a graduated cylinder by 35.80 ml (b) a 'heavy syrup' made by dissolving 4.00 cups (200 g/cup) of sugar in 5.0 cups (236.6 mL/cup) to make 7.00 cups of syrup. The density of water is approximately 1 gram/ cubic centimetre (1 g/cm3). The solution is nothing more than using conversion factors to convert grams/liter (g/L) to lbs/gal. Some factors which affect the density of water are given in the points below. the row/column shaded in aquamarine shows the density of pure water at 17.7☌ 0.998650 grams/cm 3 Density of Water (g/cm 3) at Temperatures from 0☌ (liquid state) to 30.9☌ by 0.1☌ inc. Q: I am trying to explain to my laboratory technology students the mathematical reason that when using a 83.2 cm3 weight to volume cup, just multiplying the grams of the substance by 0.1 will give you the lbs/gallon.Ī: This is a great question! I've always wanted to attack this problem myself and now you have given me the excuse to do so. Well, grams are equal to ml only if the density of the product is equal to 1 gram/cm³ ( 1 kg/l 1000 kg/m³ 62.428 lb/cu ft check this value in other units with our density converter). The density of pure water is a constant at a particular temperature, and does not depend on the size of the sample. Ask the Expert Question-and-Answer ArchiveĬonverting Grams/Liter to Lbs/Gal Using a Density (WPG) Cup 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
